The new reality in osteoarthritis care
Newsletter for physicians interested in regenerative orthopedics, March 2026
The treatment of knee osteoarthritis is at a turning point. For years, everything revolved around rapid symptom management: slightly less pain, slightly less swelling, and then hoping the joint would remain quiet for the time being. However, it is becoming increasingly clear that this strategy does not align with what we now know about osteoarthritis biology. Sustainable results are not achieved by dampening symptoms, but through interventions that structurally support the joint and actually improve the synovial environment.
Yet, we see a striking paradox in daily practice. While guidelines advise being cautious with corticosteroids, they are still used on a massive scale in the Netherlands — in 2024 alone, over 70,000 knee injections were administered. Caution? Or simply the result of insured care, in which treatments that actually align with modern joint preservation often fall outside of reimbursement?
In any case, the literature is increasingly clear: repeated use of corticosteroids has little place in contemporary osteoarthritis treatment, except in exceptional situations. And that is not because there are no alternatives — on the contrary. In recent years, therapies have been developed that precisely meet the needs of an osteoarthritic joint: restoration of synovial balance, inhibition of inflammation, and support of natural biomechanics.
In this newsletter, we zoom in on two of those options: hyaluronic acid and Arthrosamid®. Hyaluronic acid is often underestimated, even though its effectiveness is highly dependent on molecular weight, dosage, and CD44 activation. Additionally, we discuss how the synovium — long viewed as a passive lining — actually plays a central role in pain, inflammation, and the progression of osteoarthritis, and how Arthrosamid®, as a synovial implant, intervenes precisely there.
Enjoy the read,
Paul
Paul van der Wielen MSc BSc
Supportho Medical BV.
Mobile +31653256063 (WhatsApp)
Email paul@supporthomedical.nl
Corticosteroids in one minute
Newsletter for physicians interested in regenerative orthopedics, March 2026
Corticosteroids have held a steady place in osteoarthritis care for years, primarily due to their rapid analgesic effect. However, modern literature paints a different picture. The well-known JAMA study by McAlindon demonstrates this clearly: patients who received intra-articular triamcinolone every 3 months lost significantly more cartilage over two years than the placebo group — without any superior improvement in pain. MRI images even confirmed accelerated structural decline.
This means that the temporary relief provided by corticosteroids comes at a price: acceleration of cartilage degradation. And that is precisely why they only fit very selectively into a modern treatment path aimed at joint preservation. Especially in an era where regenerative and structural interventions — such as high-molecular-weight hyaluronic acid or synovial implants like Arthrosamid® — do contribute to synovial stability and biological quiescence.
In short: corticosteroids can help in the short term but harm in the long term. In regenerative orthopedics, their role is therefore shifting from routine injection to exception, appropriate only when other options are temporarily unavailable.
1. Corticosteroids: guideline vs. reality
Newsletter for physicians interested in regenerative orthopedics, March 2026
These developments mark a new phase in osteoarthritis care: moving away from temporary suppression toward biological stability and structural support.
Although corticosteroids are still mentioned in the Hip and Knee Osteoarthritis Guideline for short-term relief, recent literature compels caution.
Effect of Intra-articular Triamcinolone vs Saline on Knee Cartilage Volume and Pain in Patients With Knee Osteoarthritis A Randomized Clinical Trial
Timothy E. McAlindon et al.; JAMA. 2017;317(19):1967-1975. doi:10.1001/jama.2017.5283
CONCLUSIONS AND RELEVANCE Among patients with symptomatic knee osteoarthritis, 2 years of intra-articular triamcinolone, compared with intra-articular saline, resulted in significantly greater cartilage volume loss and no significant difference in knee pain. These findings do not support this treatment for patients with symptomatic knee osteoarthritis.
In summary:
- Repeated injections (every 3 months for 2 years) caused significantly more cartilage loss.
- There was no long-term superior analgesic effect compared to placebo.
- MRI showed structural decline despite temporary relief.
Conclusion: Corticosteroids can accelerate cartilage degradation and therefore only fit selectively into a modern treatment path.
Hyaluronic acid in one minute
Newsletter for physicians interested in regenerative orthopedics, March 2026
Hyaluronic acid is no longer just a simple “lubricant.” Modern science shows that it is primarily a biological modulator that temporarily returns the osteoarthritic joint to a healthier state. After injection, lubrication improves immediately, but the real work happens at the cellular level: the long HA chains bind to CD44 receptors, inhibiting inflammatory genes such as IL-1β, TNF-α, and IL-6, and reducing the activity of degradative enzymes (MMPs, ADAMTS). Simultaneously, synoviocytes are stimulated to produce endogenous hyaluronic acid again — an anabolic reset that continues to work for weeks to months.
A prerequisite for this effect is quality: high molecular weight (>1.5–4 MDa) for strong CD44 clustering, and a sufficiently high dosage (>60 mg) to achieve both receptor occupancy and a longer residence time in the joint. Although exogenous HA is broken down within a few weeks, the biological message persists much longer. This explains why a single injection often provides 6–12 months of pain reduction and improved function.
In short: hyaluronic acid works not because it stays, but because it reprograms the joint — thereby temporarily breaking the vicious cycle of osteoarthritis.
2. Hyaluronzuur bij knie‑artrose: het verrassende verhaal achter een oude bekende
Newsletter for physicians interested in regenerative orthopedics
We have known hyaluronic acid for years. It was long known as the “lubricant” we injected into a squeaky joint to make things run a bit smoother. But anyone following recent literature sees that this image is now hopelessly outdated. The real story is much more interesting — and frankly, much more logical.
A joint that locks itself
Osteoarthritis is not wear and tear in the classical sense. It is a joint that loses its balance. As soon as inflammatory substances such as IL-1β and TNF-α take control, the synovium turns into a kind of “alarm center” that continuously sends out new signals. The body’s own hyaluronic acid becomes thinner and shorter, causing the synovia to lose its resilience.
And then the inevitable happens: less shock absorption → more stress → more inflammation → even less HA.
The joint enters a vicious cycle that continues to feed itself.
What happens as soon as you inject hyaluronic acid
The first few minutes are predictable: the synovia thickens, lubrication improves, friction decreases. But that is only the mechanical prologue.
The real story begins when the HA chains reach the CD44 receptors — small proteins on synoviocytes and chondrocytes that act as biological switches.
Anti-inflammatory effect
As soon as HA binds to CD44, it dims the production of IL-1β, IL-6, TNF-α, and PGE2. The joint literally comes to rest.
Inhibition of degradative enzymes
MMPs and ADAMTS — the enzymes that break down cartilage — become less active. It is as if you are putting the “demolition crew” on pause for a moment.
Stimulation of synthesis
Synoviocytes start producing endogenous hyaluronic acid again. Chondrocytes produce more proteoglycans. The joint receives a biological recovery moment it hasn’t had in a long time.
And all of this through a molecule we once primarily saw as a lubricant.
Why molecular weight and dosage matter so much
Not every hyaluronic acid can tell this story. It revolves around two crucial properties: length and quantity.
- High molecular weight (>1.5–4 MDa)
Long HA chains (high molecular weight) can grasp multiple CD44 receptors simultaneously. That sounds technical, but it simply means the signal to the cell becomes stronger and more stable. Short fragments (low molecular weight) do not do this — in fact, they can even have a pro-inflammatory effect.
- High dosage (>60 mg)
A higher dose ensures that:
- there is enough HA present to activate sufficient CD44 receptors
- the lymphatic system becomes temporarily “saturated,” allowing the HA to remain in the joint longer
- enzymes such as hyaluronidase cannot break everything down immediately
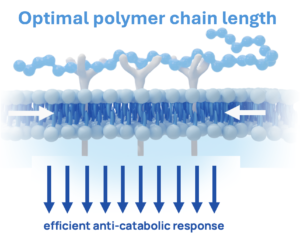
It is therefore not a matter of “filling more,” but of crossing biological thresholds. In this regard, a very high dose of 120 mg is likely recommended in a single or multiple treatments.
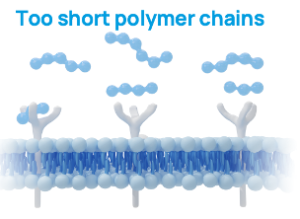
Too short: Catabolic response
Optimal: Efficient anti-catabolic response
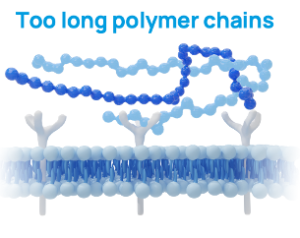
Too Long: Not optimal anti-catabolic response
Linear versus cross-linked: two forms with their own personality
Linear HA
This is the fast communicator. The chains move freely, bind efficiently to CD44, and provide a powerful biological stimulus. The mechanical half-life is shorter, but with a high dosage, you can achieve a strong cumulative effect and extend the half-life as a result of “saturation.”
Cross-linked HA (BDDE)
This is the slow, stable force. Due to its 3D structure, the material remains present for weeks, providing long-term shock absorption and biological stimulus. The downside: up to 20% of patients experience a temporary inflammatory reaction — unpleasant, but self-limiting.
Both forms have their place, as long as you know which effect you want to achieve.
Why does hyaluronic acid work for 6–12 months while it disappears within days to weeks?
This is perhaps the most intriguing part of the story.
Although exogenous HA is broken down within a few days to weeks, the biological message persists much longer. CD44 activation sets processes in motion that do not simply stop:
- NF-κB remains suppressed
- MMPs remain inhibited
- synoviocytes continue to produce more endogenous HA
- chondrocytes continue to build proteoglycans
The joint functions as if it has been temporarily “reprogrammed.” This is the remanent anabolic signal — the reason patients experience less pain and better function for months.
What does this mean for clinical practice?
When you use hyaluronic acid, you are not just giving the joint a little extra lubrication. You are temporarily breaking the vicious cycle of osteoarthritis and creating a biological window in which recovery becomes possible again.
- Choose high molecular weight to optimally activate CD44.
- Dose >60 mg, ideally higher, to reach the biological threshold.
- Expect 6–12 months of clinical benefit — not from the material itself, but from the cellular reset it causes.
- Use this period to guide patients toward GLA:D or other strength/stabilization training combined with lifestyle advice, so that the mechanical benefit also persists in the long term.
References:
- CD44 binding & cytokine inhibition
- Campo GM, Avenoso A, Campo S, et al. Hyaluronan mediates anti-inflammatory effects via CD44. Arthritis Res Ther. 2009;11(6):R166.
- Glinkowski WM, Narloch J, Glinkowska B. Molecular mechanisms and therapeutic role of intra-articular hyaluronic acid in osteoarthritis. J Clin Med. 2025;14(3):512.
- Yasuda, T. (2011). Hyaluronan inhibits cytokine production by blocking JNK and NF-κB activation via CD44. Inflammation Research, 60(6), 541-548.
- Litwiniuk, M., et al. (2016). Hyaluronic Acid in Inflammation and Tissue Regeneration. Wounds, 28(3), 78-88. Wang, C. T., et al. (2006). High molecular weight hyaluronan inhibits IL-1β-induced inflammation in cartilage chondrocytes. Osteoarthritis and Cartilage, 14(12), 1239-1247.
- R. Altman et al., Anti-Inflammatory Effects of Intra-Articular Hyaluronic Acid: A Systematic Review. Cartilage 2019, Vol. 10(1) 43–52
- Inhibition of MMPs, ADAMTS & stimulation of endogenous HA
- Ghosh P, Guidolin D. Mechanisms of HA in OA. Semin Arthritis Rheum. 2002;32(1):10-37.
- Altman RD, Manjoo A, Fierlinger A, et al. Mechanism of action of hyaluronic acid in OA: systematic review. BMC Musculoskelet Disord. 2015;16:321.
- Julovi, S. M., et al. (2004). Inhibition of interleukin-1β-induced effector functions in human articular chondrocytes by low- and high-molecular-weight hyaluronan. Arthritis & Rheumatism, 50(2), 516-525.
- Monfort, J., et al. (2008). Biochemical biomarkers in osteoarthritis: focuses on biological aspects of hyaluronan. ScienceDirect/Arthritis Research & Therapy.
- Higher molecular weight = better clinical response
- Hummer CD, et al. High molecular weight HA for knee OA: network meta-analysis. BMC Musculoskelet Disord. 2020;21:702.
- Henrotin Y, Raman R, Richette P. Consensus on viscosupplementation. Semin Arthritis Rheum. 2015;45(2):140-149.
- Dosing principles
- Altman RD. Intra-articular hyaluronic acid: dosing considerations. Osteoarthritis Cartilage. 2015;23(3):363-365.
- Conrozier, T., et al. (2016). Is arthroscopic synovial fluid dilution a real problem? Focus on viscosupplementation dosage. Journal of Clinical Medicine.
- Half-life & residence time in the joint. Linear HA (10–24 hours, effect longer)
- Kobayashi S, et al. Pharmacokinetics of intra-articular hyaluronic acid. Clin Pharmacokinet. 2004;43(8):551-566.
- Crosslinked HA (days–weeks)
- Synvisc (Hylan G-F 20) pharmacokinetics. Data summarized in: Moreland LW. Rheumatology. 2003;42(9):1239-1248.
- Durolane (NASHA) half-life. Summarized in: Henrotin Y, et al. Semin Arthritis Rheum. 2015;45(2):140-149.
- Crosslinked HA & post-injection reactions (up to 20%)
- Puttick MP, Wade JP, Chalmers A, et al. Acute local reactions after hylan G-F 20. J Rheumatol. 1995;22(7):1311-1314.
- Waddell DD, Bricker DC. Severe acute localized reactions to hylan G-F 20. Clin Rheumatol. 2007;26(11):1973-1976.
- Remanent anabolic signal (long-term effect despite short half-life)
- Long-term modulation of cytokines & MMPs
- Campo GM, et al. CD44-mediated anti-inflammatory cascade. Arthritis Res Ther. 2009;11(6):R166.
- Glinkowski WM, et al. Long-term biological modulation by HA. J Clin Med. 2025;14(3):512.
- Effects persist for months
- Ghosh P, Guidolin D. Biological persistence of HA effects. Semin Arthritis Rheum. 2002;32(1):10-37.
- Bellamy N, Campbell J, Robinson V, et al. Viscosupplementation for knee OA. Cochrane Database Syst Rev. 2006;(2):CD005321.
- Henrotin Y, et al. Consensus on long-term clinical benefit. Semin Arthritis Rheum. 2015;45(2):140-149.
- Bagga, H., et al. (2006). Long-term effects of intra-articular hyaluronan on synovial fluid viscosity and its correlation with clinical outcome. Rheumatology, 45(1), 94-98.
- Long-term modulation of cytokines & MMPs
- General
-
- Pavan Walvekar et al., A review of hyaluronic acid-based therapeutics for the treatment and management of arthritis. International Journal of Biological Macromolecules 264 (2024) 130645
- Krzysztof Falkowski, Single Injection of Highly Concentrated Hyaluronic Acid Provides Improvement of Knee Joint Arthrokinematic Motion and Clinical Outcomes in Patients with Osteoarthritis—Non-Randomized Clinical Study. J. Clin. Med. 2025, 14, 3557, 2-14
- Julian B. Troncoso et al., Different hyaluronan concentration determinate distinctive physicochemical and biological properties of the intra-articular gel but does not affect satisfactory clinical outcome. Journal of Orthopaedic Reports 4 (2025) 100445
-
Arthrosamid® in one minute
Newsletter for physicians interested in regenerative orthopedics, March 2026
Arthrosamid® is not an ordinary injection, but an injectable synovial implant. After administration, the soft hydrogel slowly intertwines with the synovium, making the capsule thicker, more flexible, and less painful. It feels as if the joint is structurally improving from the inside out.
But the story goes beyond mechanics. Perhaps the most striking effect is that the synovium becomes up to 25% more elastic once the hydrogel is fully integrated — as if the joint is fitted with a smoother, more resilient coat. Simultaneously, something happens that you don’t see on an ultrasound but do find in the biology: the gel “reprograms” the synovium. Inflammatory genes such as IL-1β and TNF-α are dampened, recovery genes are activated, and the barrier function of the capsule becomes stronger and less permeable. Patients with active synovitis particularly benefit from this; it is precisely in that overactive, inflamed environment that the gain is greatest and the unique action of Arthrosamid® comes into its own.
The result is remarkably durable: many patients experience years of pain reduction, better function, and a significantly lower risk of a knee prosthesis. The effect builds up slowly, peaking between 3 and 6 months, but often lasts up to 5 years.
In short: Arthrosamid® is an injection that behaves like an implant — and structurally improves the joint from the inside out.
3.Arthrosamid®: the story of an injection that is not an injection, but an implant
Newsletter for physicians interested in regenerative orthopedics, March 2026
Sometimes a treatment comes along that doesn’t fit neatly into an existing category. Arthrosamid® is exactly such a treatment. Not a corticosteroid, not hyaluronic acid, not PRP — but a completely new class: an injectable synovial implant. That sounds almost paradoxical: an implant that you inject through a needle. But anyone who understands what happens in the joint afterward sees why this term is exactly right.
How a hydrogel changes the synovium
When you inject Arthrosamid, it doesn’t stay floating as a loose gel in the joint cavity. It seeks contact with the synovium — and there begins a process that is surprisingly biological.
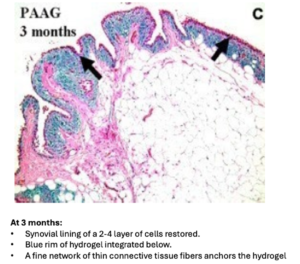
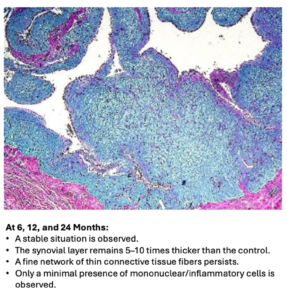
Within a few months, the gel is fully integrated into the synovial membrane. Not as a hard plug, but as a soft, flexible network that becomes interwoven with the synovial fibers. Animal studies and human data show the same pattern:
- the synovium becomes 5–10× thicker
- elasticity increases by 15–27%
- the pathological stiffness of osteoarthritis normalizes
It is as if the capsule receives a new layer that better distributes mechanical load. Not by pushing pressure away, but by reducing tissue deformation — a subtle but important difference.
How do we know that?
Because three completely different research methods show the same thing:
- Mathematical simulations show that the gel itself carries hardly any tension, thereby relieving the synovium.
- Rheometry shows that stiff, osteoarthritic membranes regain the flexibility of healthy joints.
- Sono-elastography confirms that human synovia actually become more elastic after 3–4 months.
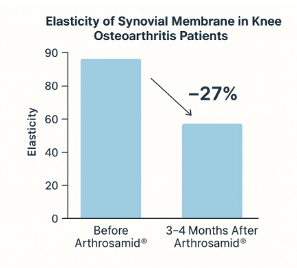
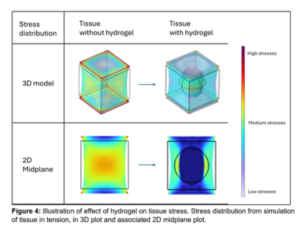
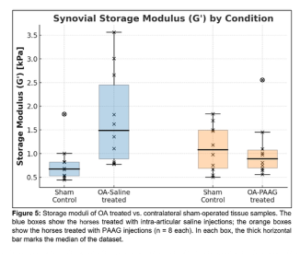
The synovium thus not only becomes thicker — it becomes functionally better.
From mechanical support to genetic reprogramming
The most intriguing part of the story takes place at the cellular level. Arthrosamid is not a passive gel; it influences the gene expression of synoviocytes.
Research by Pezzanite, Snow, and others shows that iPAAG (2.5% polyacrylamide hydrogel):
- IL‑1β and TNF‑α suppressed
- matrix repair genes activated
- the barrier function of the synovium strengthened
The synovium becomes less “leaky,” effusion decreases, and the natural filter function is restored.
Even the synovial fluid changes:
- IL-1RA rises after 3 months
- IL-6 decreases
- endogenous hyaluronic acid increases
The joint behaves as if it has been biologically “reset” to a healthier state.
Why patients with synovitis actually respond better
A striking finding presented by Prof. Snow during ISAKOS 2025: patients with active synovitis have a higher chance of being ‘responders.’
That goes against our classical thinking, but biologically it makes sense.
- A ‘target-rich environment’
In active synovitis, IL-1β and TNF-α are strongly elevated. It is precisely those genes that are suppressed by iPAAG. The more active the inflammation, the greater the gain.
- Restoration of a leaky barrier
Inflamed synovium is often hyperpermeable. Arthrosamid restores shear stiffness and, as it were, seals the “cracks” in the capsule. Patients feel this immediately as less swelling and less pressure pain.
- The greatest gain is where the greatest abnormality lies
A quiet, extinguished osteoarthritic joint has less room for improvement. An actively inflamed joint does — and therefore responds more strongly.
This makes Arthrosamid a precision intervention, not a last resort.
Clinical results: a treatment that continues to work for years
The long-term data are perhaps the most impressive:
- 80% of patients <70 years with grade ≥2 osteoarthritis show significant improvement
- effect lasts up to 5 years after a single injection
- in a 10-year follow-up, 56% still did not require a knee prosthesis
- WOMAC and KOOS scores improve by an average of 20 points
Safety first!
And all of this with a safety profile built over 25 years in more than 1.8 million treatments (including for stress incontinence).
Side effects are usually mild: some swelling, some post-injection pain — but no serious product-related complications.
The Arthrosamid package leaflet advises applying antibiotic prophylaxis. Arthrosamid is an injectable implant and must be treated as such.
The integration phase: the story you should tell patients
Arthrosamid does not work like a corticosteroid or hyaluronic acid. The effect builds up slowly because the gel needs time to integrate into the synovium.
The most important message for patients:
- Some patients have an immediate effect after treatment. After a few days to weeks, pain symptoms may increase again.
- the first effects begin around 6 weeks
- maximum effect occurs between 3 and 6 months
- the result is an average of 20 points improvement — but that is an average
Some patients are virtually pain-free. Others improve moderately. A small group does not respond.
Good expectation management is therefore essential.
What does this mean for your practice?
Arthrosamid is not a lubricant, not an anti-inflammatory, and not a regenerative therapy. It is a synovial implant that:
- structurally changes the synovium
- modulates the gene expression of synoviocytes
- restores barrier function
- normalizes joint mechanics
- and thereby provides years of clinical benefit
It is a treatment that not only dampens symptoms but changes the joint environment itself.
References:
- Intra-articular 2.5% polyacrylamide hydrogel, a new concept in the medication of equine osteoarthritis: A review; Tnibar A. Equine Vet Sci. 2022; 119: 104143
Link: https://www.sciencedirect.com/science/article/abs/pii/S0737080622002799 - Synovial incorporation of polyacrylamide hydrogel after injection into normal and osteoarthritic animal joints; Christensen, L, et al. Osteoarthritis Cartilage. 2016; 24 (11): 1999-2002
Link: https://www.oarsijournal.com/action/showPdf?pii=S1063-4584%2816%2930189-3 - An international multi-centre prospective study on the efficacy of an intraarticular polyacrylamide hydrogel in horses with osteoarthritis: a 24 months follow-up; Tnibar A, et al. Acta Vet Scand. 2015; 57 (1) Link: https://pmc.ncbi.nlm.nih.gov/articles/PMC4403890/pdf/13028_2015_Article_110.pdf
- Use of a 2.5% Cross-Linked Polyacrylamide Hydrogel in the Management of Joint Lameness in a Population of Flat Racing Thoroughbreds: A Pilot Study; de Clifford LT, et al. J Equine Vet Sci. 2019; 77: 57-62 Link: https://www.sciencedirect.com/science/article/abs/pii/S0737080618307615
- Intra-articular 2.5% polyacrylamide hydrogel for the treatment of knee osteoarthritis: an observational proof-of-concept cohort study;1Henriksen M, et al. Clin Exp Rheumatol. 2018; 36 (6): 1082-1085 Link: https://pubmed.ncbi.nlm.nih.gov/30148430/
- 6 Months – Polyacrylamide Hydrogel Injection for Knee Osteoarthritis: A 6 Months Prospective Study. Bliddal H, Overgaard A, Hartkopp A, et al. J; Orthop Res Ther 6. 2021; 1188. Link: doi: 10.29011/2575-8241.001188
- 12 Months –Polyacrylamide Hydrogel Injection for Knee Osteoarthritis: Results of a 52 Week Prospective Study. Bliddal H, Overgaard A, Hartkopp A, et al. Abstract, Osteoarthritis and Cartilage. 2021; 29: S278. Link: doi: 10.1016/j.joca.2021.02.366
- 12 months – Effectiveness and safety of polyacrylamide hydrogel injection for knee osteoarthritis: results from a 12-month follow up of an open-label study. Bliddal H, et al. Journal of Orthopaedic Surgery and Research Surg Res 2024;19, 274. Link: doi: 10.1186/s13018-024-04756-2
- 24 Months –A Prospective Study of Polyacrylamide Hydrogel for Knee Osteoarthritis: Results from 2 years After Treatment. Bliddal H, Beier J, Hartkopp A, et al. Abstract, Osteoarthritis and Cartilage. 2022; 30: S372. Link: doi: 10.1016/j.joca.2022.02.499
- 36 Months –3 Year Results from a Prospective Study of Polyacrylamide Hydrogel for Knee Osteoarthritis. Abstract, Osteoarthritis and Cartilage. Henriksen M, Beier J, Hartkopp A, et al. 2023; 31: 682-683. LINK:doi: 10.1016/j.joca.2023.02.023
- 4-years- POLYACRYLAMIDE HYDROGEL FOR KNEE OSTEOARTHRITIS: 4-YEAR RESULTS FROM A PROSPECTIVE STUDY; Henning Bliddal, EORS 2024, Oral presentation abstract. www.EORS2024.org
Link: chrome-extension://efaidnbmnnnibpcajpcglclefindmkaj/https://eors2024.org/wp-content/uploads/2024/09/all_oral_abstracts.pdf - Polyacrylamide Hydrogel for Knee Osteoarthritis: 5-Year Results from a Prospective Study; Bliddal et al. ;Presented at WCO-IOF-ESCEO 2025.
Link: https://www.oarsijournal.com/article/S1063-4584(22)00533-7/fulltext - One year Performance of Polyacrylamide Hydrogel vs. Hyaluronic Acid: A Randomised Controlled Study. Bliddal H, Beier J, Hartkopp A, et al. Abstract, Osteoarthritis and Cartilage. 2022; 30: 371.
Link: doi: 10.1016/j.joca.2022.02.497 - Polyacrylamide gel versus hyaluronic acid for the treatment of knee osteoarthritis: A randomised controlled study. Bliddal H, Beier J, Hartkopp A, et al. Clinical and Experimental Rheumatology. 2024 Mar 19. Link: org/10.55563/clinexprheumatol/i3fqee Epub ahead of print. PMID: 38525999
- Histological Appearance of the Synovial Membrane after Treatment of Knee Osteoarthritis with Polyacrylamide Gel Injections: A Case Report. Christensen LH, Daugaard S. J Arthritis. 2016; 5: 5.
Link: doi: 10.4172/2167-7921.100021 - Reduction in Patellofemoral Bone Marrow Lesions Following Single Arthrosamid Intra-Articular Injection of Polyacrylamide Hydrogel (iPAAG) in the Treatment of Advanced Osteoarthritis. Maulana R, Cole A, Lee PYF. Journal of Arthritis. 2022; 11 (3): 024-026
Link: https://www.theregenerativeclinic.co.uk/wp-content/uploads/2022/07/Treatment-of-Advanced-Osteoarthritis_P.Lee_.pdf#:~:text=We%20present%20the%20first%20case%20of%20a%20reduction,presented%20to%20the%20clinic%20with%20anterior%20knee%20pain. - Review, A Systematic Review of the Novel Compound Arthrosamid Polyacrylamide (PAAG) Hydrogel for Treatment of Knee Osteoarthritis. Cole A, Maulana R, Whitehead J.P, Lee. Medical Research Archives. 2022; 10 (8) Link: https://esmed.org/MRA/mra/article/view/2950/193546227
- 10-Year Safety Follow-Up of Intra-Articular 2.5% Polyacrylamide Hydrogel (iPAAG) in Knee Osteoarthritis (OA)Presented at: WCO-IOF-ESCEO 2025, Rome
Authors: H. Bliddal, et al. Link:https://wco-iof-esceo.org/download/2025/abstract-book page 58 OC11 - A Mechanistic and Clinical study of intra-articular Arthrosamid for knee osteoarthritis Presented at the ISAKOS congress 2025
Author(s): Sharon Owen, et al. Link: https://isakos.com/GlobalLink/Abstract/8064 - Polyacrylamide Hydrogel for Knee Osteoarthritis: 5-Year Results from a Prospective Study
Presented at WCO-IOF-ESCEO 2025.
Authors: H. Bliddal, et al.
Link: https://www.arthrosamid.nl/meer-te-weten-komen-over-de-gegevens/ (copy from abstract book) - Polyacrylamide hydrogel injections in knee osteoarthritis: A PROMs-based 24 month cohort study. Author(s) Hiu Ching Kelvin Gao et al. (Oct 2025)
Link: https://www.sciencedirect.com/science/article/abs/pii/S0976566225002346 - A SINGLE INJECTION OF INTRA-ARTICULAR ARTHROSAMID FOR KNEE OSTEOARTHRITIS – A STUDY OF CLINICAL EFFICACY AND MECHANISM; Author(s); M. Snow, et al.
Link: https://www.oarsijournal.com/article/S1063-4584(25)00792-7/abstract; Link to text - Intra-articular Arthrosamid® injection for knee osteoarthritis: A synovial fluid biomarker study
Author(s); K.T. Wright, et al.
Link: https://www.sciencedirect.com/science/article/abs/pii/S1063458425002390; Link to text - THE IMPACT OF PATIENT FACTORS ON THE MINIMAL CLINICALLY IMPORTANT DIFFERENCE OF ARTHROSAMID POLYACRYLAMIDE HYDROGEL INJECTION FOR KNEE OSTEOARTHRITIS: A COHORT STUDY
Author(s); Hiu Ching Kelvin Gao, et al.
Link: https://www.oarsijournal.com/article/S1063-4584(25)00673-9/abstract; Link to text. - An injectable 2.5% cross-linked polyacrylamide hydrogel (2.5 iPAAG) demonstrates no neurotoxicity in human induced pluripotent stem cells-derived iCell® GlutaNeurons.
Author(s): P.S. Walmod, et al.
Link: https://www.frontiersin.org/journals/toxicology/articles/10.3389/ftox.2025.1585430/ful - Comparative efficacy of polyacrylamide hydrogel versus hyaluronic acid and corticosteroids in knee osteoarthritis A retrospective cohort study; Bilal Aykaç et al; Medicine (2025) 104:38
Link: https://pmc.ncbi.nlm.nih.gov/articles/PMC12459573/ - Three-year follow-up from a randomised controlled trial of a single intra-articular polyacrylamide hydrogel injection in subjects with knee osteoarthritis; H. Bliddal et al.; Clinical and Experimental Rheumatology 2025.
Link: Three-year follow-up - Shear stiffness of the equine synovial membrane in osteoarthritis and the response to treatment with polyacrylamide hydrogel, Dr. Markus Wimmer, Rush University, Chicago (Rush University Medical Center) Department of Orthopaedic Surgery – Motion Analysis & Joint Biomechanics
ICRS presentation, Boston 2025
Link: ICRS presentations Arthrosamid/ iPAAG Boston October 4-7, 2025 - Synovial transcriptomic response to intra-articular 2.5% polyacrylamide hydrogel in an equine osteoarthritis model, R. Lynn Pezzanite, Equine Orthopedics & Translational Medicine, ICRS presentation, Boston 2025
Link: ICRS presentations Arthrosamid/ iPAAG Boston October 4-7, 2025 - The Symptom- and Disease-Modifying Effects of Intra-articular 2.5% Polyacrylamide Gel in an Equine Preclinical Osteoarthrosis Model, Dr. Erin Contino, Colorado State University, Equine Sports Medicine & Rehabilitation
ICRS presentation, Boston 2025
Link: ICRS presentations Arthrosamid/ iPAAG Boston October 4-7, 2025
The whole is greater than the sum of its parts — in one minute
Newsletter for physicians interested in regenerative orthopedics, March 2026
Injections such as Arthrosamid®, hyaluronic acid, or PRP can achieve a great deal: they reduce pain, calm inflammation, and provide the joint with biological breathing room. However, their greatest strength truly comes to life when combined with a structured exercise program such as GLA:D®. An injection opens a window of opportunity: for the first time in a long while, the patient can move again without every step causing protest. And it is precisely at that moment that exercise therapy can make the difference.
GLA:D® restores what osteoarthritis has undermined for years: muscle strength, stability, proprioception, and the confidence to use the joint again. Stronger muscles become the knee’s natural shock absorbers, ensuring that the gains from the injection are not only felt but also remain sustainable.
Additionally, the program teaches patients why movement is essential for cartilage metabolism and joint health. This education ensures better self-management and less dependence on repeated medical interventions.
In short: injection therapy makes movement possible, GLA:D® makes improvement lasting. Together, they break the vicious cycle of osteoarthritis — and that is exactly why the whole is greater than the sum of its parts.
The whole is greater than the sum of its parts
Newsletter for physicians interested in regenerative orthopedics, March 2026
Why injection therapy only becomes truly powerful in combination with GLA:D®
Those who regularly treat patients with knee osteoarthritis see it time and again: an injection can do a lot — sometimes even surprisingly much — but that injection rarely tells the whole story. Arthrosamid®, PRP, hyaluronic acid… they can dampen pain, calm inflammation, and biologically reset the joint. But as soon as the patient can move better again, something arises that is at least as important as the injection itself: an opportunity to retrain the joint.
This is the synergy that the GLA:D® program is all about. Not as an “extra,” but as the logical second half of a treatment that would otherwise remain incomplete.
The ‘window of opportunity’: when the joint finally cooperates
An injection that reduces pain does more than just provide comfort. It opens a window that has been closed for many patients for years: the moment when movement is possible again without every step causing protest.
In this phase — often the first weeks to months after the injection — the joint is biologically calmer, mechanically more stable, and functionally less painful. This is exactly the moment when exercise therapy yields the highest return.
Injection therapy makes movement possible. Exercise therapy makes improvement sustainable.
Why the body needs exercise therapy, even when the pain is gone
Osteoarthritis changes not only the cartilage but also the way the entire musculoskeletal system functions. Muscles become weaker, reflexes slower, and proprioception less sharp. The body begins to compensate — often for years.
A program like GLA:D® addresses exactly those hidden consequences.
Neuromuscular control
Due to osteoarthritis, the knee “forgets” how to work together with the surrounding muscles. GLA:D® teaches the system to coordinate again: better stability, better shock absorption, and reduced peak loading.
Muscle strength as natural protection
Strong quadriceps are the best shock absorbers a knee can have. They absorb forces that would otherwise land directly on the joint.
Education as medicine
Patients learn why movement is not an enemy but a prerequisite for healthy cartilage. This insight changes behavior — and behavior determines the long term.
Why the combination works longer than the injection alone
The literature is remarkably unanimous on this: patients who remain active maintain their gains longer.
This is logical. An injection changes the joint environment. Exercise therapy changes the way the joint is used.
Together, they break the vicious cycle of osteoarthritis:
- less pain → more movement
- more movement → better muscle function
References:
www.gladinternational.org/ www.gladnederland.nl
International guidelines (core: exercise therapy, education, lifestyle)
- OARSI 2019 – Non-surgical management of knee, hip and polyarticular OA Bannuru RR, Osani MC, Vaysbrot EE, et al. OARSI guidelines for the non-surgical management of knee, hip, and polyarticular osteoarthritis. Osteoarthritis Cartilage. 2019;27(11):1578-1589.
- Overview of guidelines – focus on exercise, education, weight reduction Macri EM, Selles RW, Stefanik JJ, Reijman M. OARSI year in review 2023: Rehabilitation and outcomes. Osteoarthritis Cartilage. 2023;31(12):1534-1547.
- ACR/Arthritis Foundation guideline (clinical practice, conservative management) Kolasinski SL, Neogi T, Hochberg MC, et al. 2019 American College of Rheumatology/Arthritis Foundation guideline for the management of osteoarthritis of the hand, hip, and knee. Arthritis Care Res. 2020;72(2):149-162.
- EULAR recommendations for knee and hip osteoarthritis Fernandes L, Hagen KB, Bijlsma JWJ, et al. EULAR recommendations for the non-pharmacological core management of hip and knee osteoarthritis. Ann Rheum Dis. 2013;72(7):1125-1135.
- Dutch context – KNGF guideline for hip/knee osteoarthritis KNGF Guideline Osteoarthritis hip-knee. Royal Dutch Society for Physical Therapy, latest update (consult KNGF site for current version).
Exercise therapy: effect on pain and function
- Therapeutic exercise for hip/knee osteoarthritis (Lancet) Fransen M, McConnell S, Harmer AR, et al. Exercise for osteoarthritis of the knee: a Cochrane systematic review. (Related: moderators of effect in The Lancet). The OARSI year in review summarizes that exercise therapy has a small but consistent positive effect on pain and function.
- Therapeutic exercise – effect size and clinical relevance Bennell KL, Hinman RS. Exercise as a treatment for osteoarthritis. Curr Opin Rheumatol. 2015;27(3):304-311.
Lifestyle and weight reduction
- Diet + exercise for knee osteoarthritis and overweight Messier SP, Mihalko SL, Legault C, et al. Effects of intensive diet and exercise on knee joint loads, inflammation, and clinical outcomes among overweight and obese adults with knee osteoarthritis. JAMA. 2013;310(12):1263-1273.
- Weight reduction as a core component of conservative treatment Summarized in OARSI guidelines and OARSI year in review 2023: weight reduction + exercise therapy as “core treatment” for overweight/obesity and hip/knee osteoarthritis.
GLA:D® and structured exercise programs
- GLA:D® Denmark – implementation and outcomes in daily practice Skou ST, Roos EM. Good Life with osteoArthritis in Denmark (GLA:D™): evidence-based education and supervised neuromuscular exercise delivered by certified physiotherapists nationwide. BMC Musculoskelet Disord. 2017;18:72.
- GLA:D® – real-world data on pain, function, and self-management Skou ST, Rasmussen S, Laursen MB, et al. The efficacy of GLA:D® in patients with knee and hip osteoarthritis: cohort data from clinical practice. (Various publications; frequently cited in guidelines and reviews such as OARSI year in review 2023).
Agenda 2026
Interesting congresses for regenerative medicine/orthobiologics in 2026
April 23 – 26 – OARSI, West Palm Beach (Florida, USA)
May 4-6, 2026 – EFORT, Malaga (Spain)
May 20-22 – ESSKA, Prague (Czech Republic)
October 8–10, 2026 – ICRS Summit, Porto (Portugal)
October 8–10, 2026 – Enorm Health, Leuven (Belgium)
October 15-16, 2026 – ISAKOS, Brussels (Belgium)
October 20-23, 2026 – DKOU, Berlin (Germany)
In our next newsletter, PRP and targeted PRP for the treatment of osteoarthritis will be highlighted.
If you have a good story from your practice or interesting scientific research in the field of osteoarthritis and/or muscle and tendon complaint treatments, please let us know so we can include it in one of the following newsletters.
For questions, ideas, and/or comments, you can contact info@supporthomedical.nl
Colophon & Copyright
This newsletter for physicians interested in regenerative orthopedics is a publication of Supportho Medical BV. Website: www.supporthomedical.nl Email: info@supporthomedical.nl
© Supportho Medical BV. All rights reserved. No part of this publication may be reproduced, stored in an automated database, or made public — in any form or by any means, whether electronic, mechanical, by photocopying, recording, or in any other way — without prior written permission from the publisher.
Although this newsletter has been compiled with great care, no rights can be derived from its content. Supportho Medical BV accepts no liability for any inaccuracies or omissions, nor for the consequences of applying the information described in clinical practice.